A Search for Biologically Active Compounds for Potential Pharmaceutical and Agronomic Applications
Two projects from our research will be briefly presented. The first performed at Ciba-Geigy Central Research Laboratories in Basel, describes the design and synthesis of modified backbones for their incorporation into antisense oligonucleotides for potential therapeutical applications. Among the various modifications studied, the oligonucleotides incorporating the “Amide-3” backbone displayed improved thermodynamic stability of the corresponding duplexes with complementary RNA target compared to the wild type.1a) In addition, a largely increased resistance towards nucleases degradation has been observed. Furthermore, the” Amide-3” backbone has been largely improved by further preorganisation of the five-membered rings with 2’-OMe substitution and of the backbone by an additional 5-’(S) Me substituent.1b)
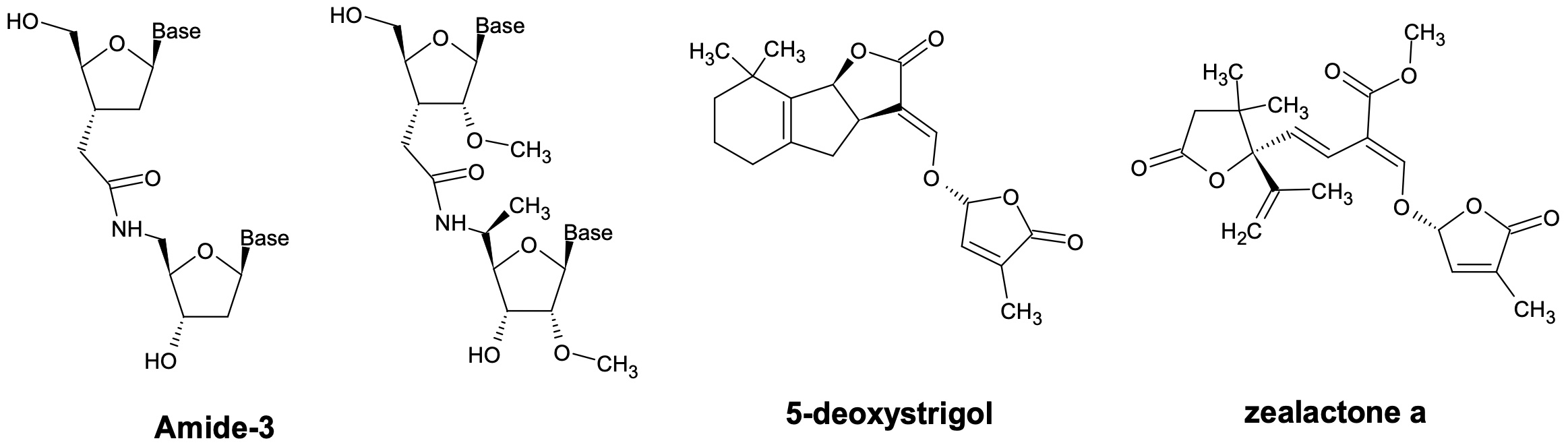
The second project, performed at Syngenta in Stein, aimed to discover improved strigolactones derivatives to stimulate plant growth and plant/soil health. Strigolactones and synthetic strigolactams strongly stimulated seed germination of crop plants under cold stress. The synthesis of several natural strigolactones as 5-deoxystrigol will be described together with their potential for Crop Enhancement.2)In particular, we identified and synthesized the major strigolactone from maize, that we named zealactone, and demonstrated its great potential for maize growth stimulation at very low rate.2b) Strigolactones and their synthetic derivatives could be very useful for sustainable agriculture.
[1] a) A. De Mesmaeker, A. Waldner, J. Lebreton, P. Hoffmann, V. Fritsch, R. M. Wolf, S. M Freier, Angew. Chem. Int. Ed. Engl. 1994, 33, 226; b) A. De Mesmaeker, C. Lesueur, M.-O. Bévierre, A. Waldner, V. Fritsch, R. M. Wolf, Angew. Chem. Int. Ed. Engl. 1996, 35, 2790.
[2] a) M. Lachia, P.-Y. Dakas, A. De Mesmaeker, Tetrahedron Lett. 2014, 55, 6577 b) M. Yoshimura, M. Dieckmann, P.-Y. Dakas, R. Fonné-Pfister, C. Screpanti, K. Hermann, S. Rendine, P. Quinodoz, B. Horoz, S. Catak, A. De Mesmaeker, Helv. Chim. Acta 2020, 103, 2000017.